States of Matter
 States of Matter Overview
States of Matter Overview
Matter is anything that has mass and occupies space. All matter is made up of tiny particles that are in constant motion. Depending on how these particles are arranged and how they move, matter exists in different physical forms called states of matter.
🔍 Particle Nature of Matter
To understand the states of matter, we must first look at the characteristics of the particles that compose it:
- Spaces: There are intermolecular spaces between particles.
- Motion: Particles are constantly moving (Kinetic Energy).
- Attraction: Particles exert a force of attraction on each other.
Changing the State
Matter can transition from one state to another by changing Temperature or Pressure.
- Temperature: Increasing heat increases particle movement and can break the forces of attraction.
- Pressure: Increasing pressure brings particles closer together, which can liquefy gases.
🔹 The Three Main States of Matter
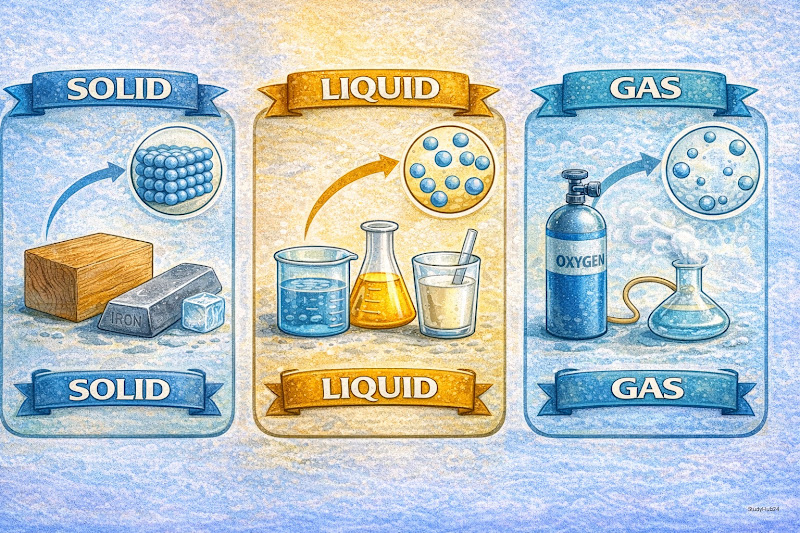
1. Solid
- Fixed Shape and Volume: Solids maintain their structure regardless of the container.
- Particle Arrangement: Particles are very tightly packed in an orderly manner.
- Force: Strong intermolecular forces of attraction.
- Examples: Ice, Wood, Iron, Diamond.
2. Liquid
- No Fixed Shape: Liquids take the shape of their container.
- Fixed Volume: They cannot be compressed easily.
- Particle Arrangement: Particles are less tightly packed than in solids and can move past each other (flow).
- Examples: Water, Oil, Milk, Mercury.
3. Gas
- No Fixed Shape or Volume: Gases expand to fill the entire volume of any container.
- High Compressibility: Because of large spaces between particles, gases can be easily compressed.
- Particle Arrangement: Particles are very far apart and move randomly at high speeds.
- Examples: Oxygen, Carbon Dioxide, Steam, Air.
📊 Properties Comparison
| Property | Solid | Liquid | Gas |
|---|---|---|---|
| Shape | Fixed | Not Fixed | Not Fixed |
| Volume | Fixed | Fixed | Not Fixed |
| Density | High | Medium | Low |
| Compressibility | Negligible | Low | Very High |
| Diffusion | Very Slow | Slow | Very Fast |
🔄 Changes of State
| Change | Process Name | Example |
|---|---|---|
| Solid → Liquid | Melting | Ice turning to water |
| Liquid → Solid | Freezing | Water turning to ice |
| Liquid → Gas | Evaporation / Boiling | Water boiling into steam |
| Gas → Liquid | Condensation | Dew forming on grass |
| Solid → Gas | Sublimation | Camphor or Dry Ice disappearing |
🌫️ The "Extra" States of Matter
Beyond the common three, scientists have identified two more states:
4. Plasma (The High-Energy State)
- Found in the Sun, stars, and lightning.
- Consists of super-heated, ionized gas that conducts electricity.
- It is the most common state of matter in the visible universe.
5. Bose-Einstein Condensate (The Low-Energy State)
- Formed when a gas of atoms is cooled to temperatures very near absolute zero (-273.15°C).
- Atoms move so slowly that they begin to behave like a single "super atom."
🧪 Fundamental Gas Laws
- Boyle’s Law: At constant temperature, the pressure of a gas is inversely proportional to its volume.
- Charles’ Law: At constant pressure, the volume of a gas is directly proportional to its absolute temperature.
- Avogadro’s Law: Equal volumes of all gases, at the same temperature and pressure, contain an equal number of molecules.
 Phase Changes
Phase Changes
📘 Practice Questions
🔹 Conceptual Check
- Why do liquids take the shape of their container?
👉 Because the intermolecular forces are not strong enough to hold particles in a fixed position, allowing them to flow. - Why does perfume smell spread quickly in a room?
👉 Due to the high rate of diffusion in gases; particles move randomly and mix with air. - Why do we feel cool when sweat evaporates?
👉 Evaporation is an endothermic process; it absorbs heat from our body to convert liquid sweat into vapor, causing a cooling effect.
🧠 Multiple Choice Questions (MCQs)
Q1: Which state of matter has the highest kinetic energy of particles?
- a) Solid
- b) Liquid
- c) Gas ✅
- d) All are equal
Q2: The process of change from gas to liquid is called:
- a) Condensation ✅
- b) Sublimation
- c) Deposition
- d) Fusion
Q3: Which of the following is highly compressible?
- a) Ice
- b) Water
- c) Oxygen ✅
- d) Wood
Q4: Diffusion is fastest in:
- a) Solids
- b) Liquids
- c) Gases ✅
- d) Plasma
Q5: What happens to the rate of evaporation as the surface area increases?
- a) It increases ✅
- b) It decreases
- c) It remains the same
- d) It stops
📖 Case Study: Sublimation
Scenario: You place naphthalene balls (mothballs) in your cupboard. After a few months, you notice they have become much smaller or disappeared entirely without leaving any liquid residue.
- Q: What process is demonstrated here?
👉 Sublimation: The direct change of a solid into gas without passing through the liquid state. - Q: Name another substance that undergoes this process.
👉 Dry Ice (Solid CO₂) or Camphor.